Hydrogen fuel cells are an energy conversion device that continuously converts the chemical energy of hydrogen and oxygen into electrical energy. Hydrogen fuel cells operate at a higher efficiency than internal combustion engines, demonstrating an electrical energy conversion efficiency of 60% or higher and having lower emissions. Water is the only product in the power generation process of hydrogen fuel cells, so there are no carbon dioxide emissions or air pollutants during operation.
Hydrogen fuel cells consist of three adjacent parts, namely the anode, electrolyte, and cathode. When hydrogen undergoes an oxidation reaction at the anode, it generates cations that migrate through the electrolyte to the cathode and free electrons that flow through the external circuit. Conversely, a reduction reaction occurs at the cathode, where oxygen is reduced to water by cations and electrons.
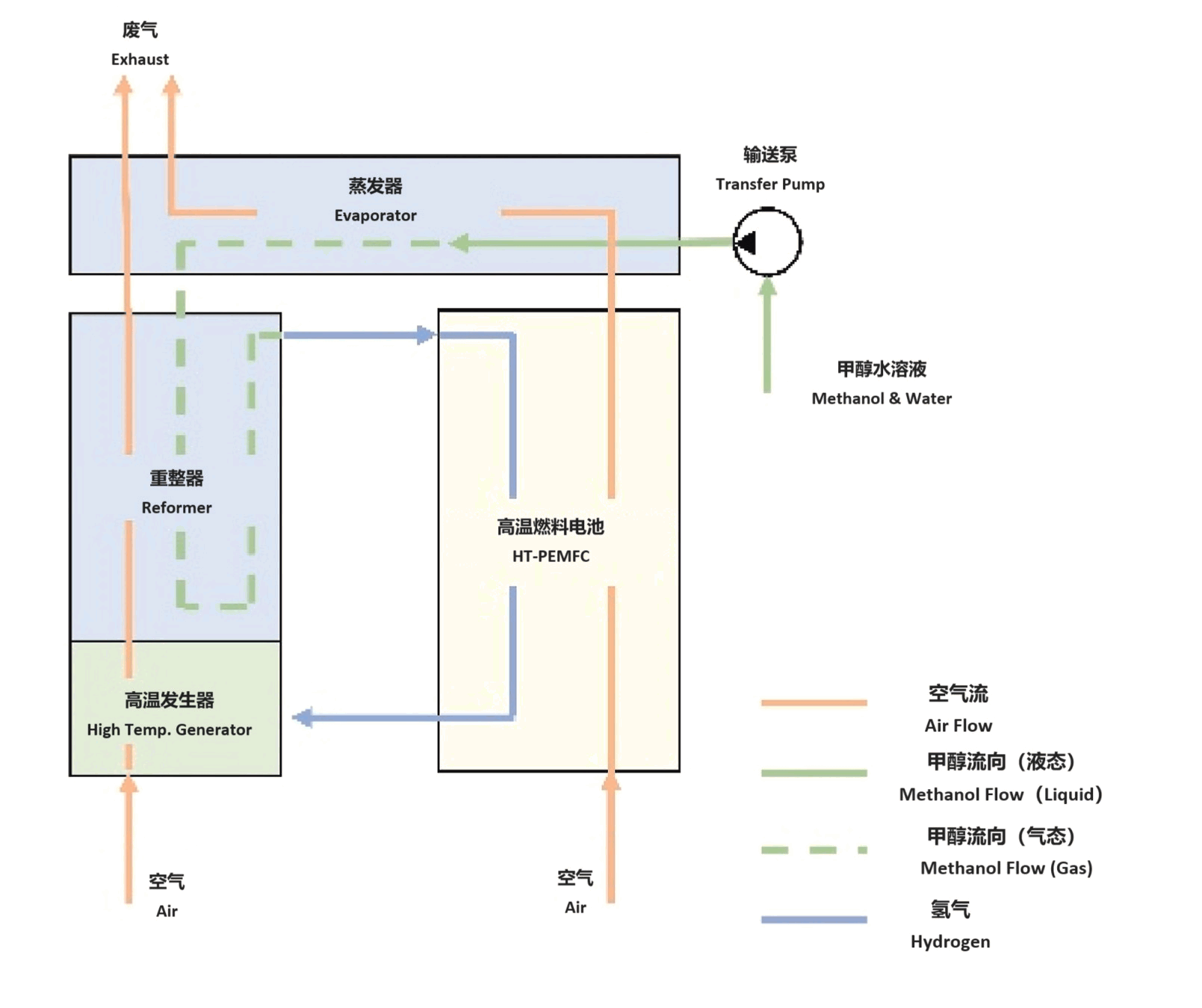
According to the type of electrolyte used, fuel cells can be classified as alkaline fuel cells (AFCs), proton exchange membrane fuel cells (PEMECs), phosphoric acid fuel cells (PAFCs), molten carbonate fuel cells (MCFCs), and solid oxide fuel cells (SOFCs). The classification and characteristics of fuel cells are listed in the following table.
| Type | AFCs | PEMFCs | PAFCs | MCFCs | SOFCs |
|---|---|---|---|---|---|
| Electrolyte | KOH | Perfluorosulfonic acid ion exchange membrane | H3PO4 | Li2CO3-K2CO3 | Y2O3-ZrO2 |
| Catalyst | Platinum/ruthenium | Platinum/ruthenium | Platinum | Nickel oxide | Nickel |
| Operating temperature | 65–220 °C | −40–90 °C | 150–200 °C | 650–700 °C | 600–1000 °C |
| Theoretical voltage | 1.18 V | 1.18 V | 1 V | 1.116 V | 1.13 V |
| System efficiency | 60%–70% | 43%–68% | 40%–55% | 55%–65% | 55%–65% |
For more information about hydrogen fuel cell technology:
https://baike.baidu.com/item/Hydrogen Fuel Cell/10964013